Artificial Intelligence Has Changed The Drug Discovery Game
Are you behind in drug discovery because you aren't using AI to its fullest potential?

AI has launched itself from the pages of science fiction and disrupted our industry.
At this point, if you aren’t using AI for drug discovery and repurposing, you’re setting yourself up to be left behind. As the pace of innovation continues to surge, hundreds of startups are centering their entire organization around AI and seeing astounding results. Pharma companies aren’t being shy either, with numerous big names quickly shifting to integrate AI into their practices.
Everyone, at every level of your organization, needs to be thinking about applying AI as a means to reduce costs, increase speed, and ensure your time and efforts are in pursuit of successful drug candidates and treatments.
- 230+ Startups using AI1
- 40+ Pharma companies using AI2
- 115+ Drug developed as a result of AI3
- $13.8B invested in companies & partnerships leveraging AI4
AI-Advancements
Over the past few decades, every facet of the drug discovery pipeline has in one way or another been altered by the move towards AI-guided approaches.
The exponential growth and improved coverage and availability of omics-datasets has pushed machine-learning-powered computational methods to become a critical tool in integrating, understanding, and utilizing these datasets to their fullest potential.
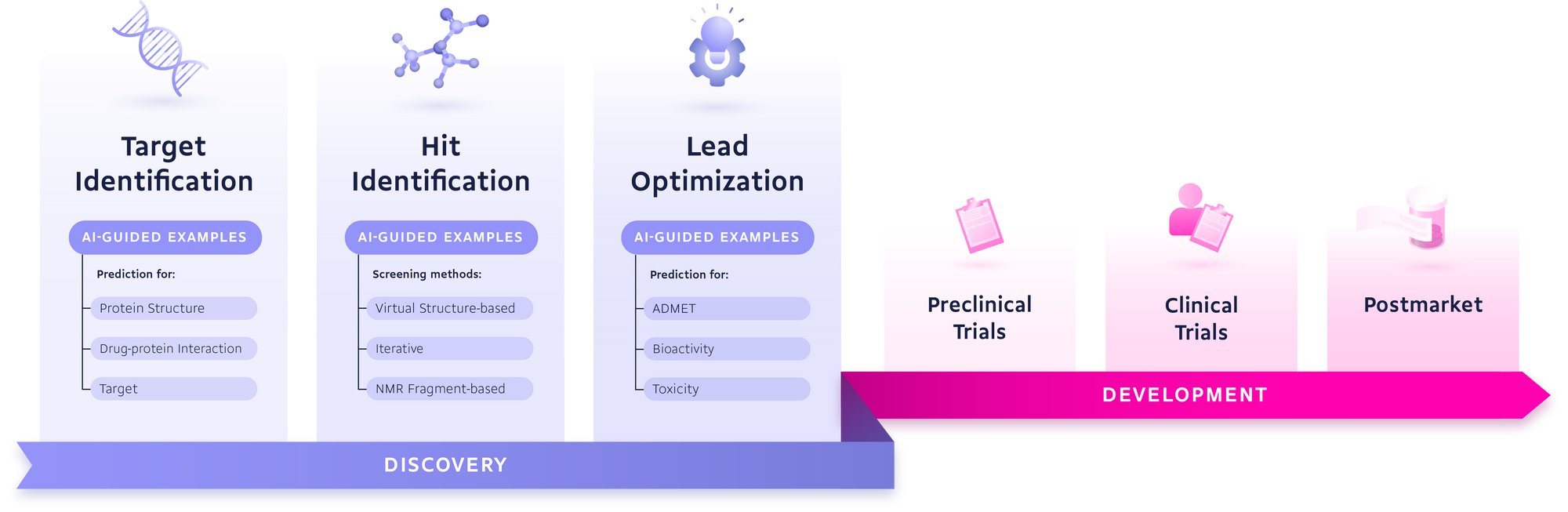
Target identification efforts have been hugely improved through the integration of varying, often heterogeneous datasets such as pathways, RNA expression, animal models, mutations, and somatic and germline genetics. The machine learning approaches that are making this possible continue to be increasingly successful in finding targets for both new and existing diseases.
The application of knowledge graphs that integrate drugs, targets, diseases, pathways, and other entities, as well as their relationships, have led to a number of promising new approaches.
One example that is still in early stages and currently seeing an intense research focus is the use of knowledge graphs to capture a more representative model of structural biology.

Big Deal Breakthroughs
Although now well-known in the drug-discovery world, the release of protein structure predictions from AlphaFold will have wide-ranging implications across the AI in drug discovery field. Beyond AlphaFold, there have also been significant advances in cryo-electron microscopy. Combining these advances introduces a new model for structural biology6, one which may well become a fertile ground for drug discovery research over the coming decade.
The gaps that exist in AlphaFold predictions (namely conformational dynamics, and disordered proteins and regions) align well with the areas where cryo-EM / cryo-ET provide useful information. The combination of AlphaFold predictions with the information on tomography being realized with cryo-EM technology7 will lead to near-atomic-resolution models of complexes, in their physiological context, inside the cell.
These advances in ML and experimental methods represent the starting point for new approaches to AI-focused drug discovery. The explosion in available high-quality 3D protein structures and the continued advancements in structured, machine-learning-ready drug data will open up new approaches, and demand entirely new optimizations to handle the intersection between the growing protein structure and chemical compound spaces.
Additionally, the COVID-19 pandemic opened the door to many new innovative, collaborative research projects focused on applying machine learning to better understand the SARS-CoV-2 virus, as well as identify novel antiviral drug candidates.
The Impact On Drug Discovery Is Undeniable
There are currently more than 100 drugs in the AI in drug discovery pipeline, and numerous drugs have been making their way to clinical trials in a matter of years—instead of decades.
As AI increasingly becomes standard practice, access to high-quality data is proving to be a true differentiator. You’ve likely heard, if not already said “garbage in, garbage out,” when it comes to drug data. As the move to AI continues to advance it will only become more vital to have reliable, clean, and robust datasets. Without this, you’ll find yourself spending more and more time getting datasets ready before you can even begin your research.
- 2M+ Scientific journals published each year8
- 7% of systemic reviews are inaccurate within 24 hours of publication9
- 23% of reviews not updated in 2 years have incorrect conclusions9
Yet, data is growing at such an exponential rate that it is becoming impossible to keep up with, maintain, structure, and normalise it at a pace and standard that enables anyone to use it to its fullest potential.
At DrugBank we work tirelessly to equip leading data scientists with the most in-depth, highest-quality, and up-to-date drug data on the market.
Download eBook
The Little Book of Big Changes in AI-Powered Drug Discovery eBook
Learn more about AI in drug discovery and get access to helpful resources and references.
References
- Smith, S. 230 Startups Using Artificial Intelligence in Drug Discovery. 2021 Jun 10.
- Smith, S. 43 Pharma Companies Using Artificial Intelligence in Drug Discovery. 2021 May 25; Accessed January 27, 2022.
- Smith, S. 116 Drugs in the Artificial Intelligence in Drug Discovery Pipeline. 2021 May 14; Accessed January 27, 2022.
- Zhang D, Mishra S, Brynjolfsson E, Etchemendy J, Ganguli D, Grosz B, Lyons T, Manyika J, Niebles JC, Sellitto M, Shoham Y, Clark J, Perrault R. The AI Index 2021 Annual Report. AI Index Steering Committee: Human-Centered AI Institute, Stanford University. 2021 March.
- Davis B. How many scientific papers are published each year? 2021 Jun 1; Accessed January 27, 2022.
- Shojania KG, Sampson M, Ansari MT, Ji J, Doucette S, Moher D. How quickly do systematic reviews go out of date? A survival analysis. Ann Intern Med. 2007 Aug 21;147(4):224-33. doi: 10.7326/0003-4819-147-4-200708210-00179. Epub 2007 Jul 16. PMID: 17638714.
- Tsao N. AI in Drug Discovery Cuts Timeline From 5+ Years to Months. 2021 Jun 10; Accessed January 27, 2022.
- Subramaniam S, Kleywegt GJ. A paradigm shift in structural biology. Nat Methods 19, 20–23. 2022 Jan 11.
- de Oliveira TM, van Beek L, Shilliday F, Debreczeni JÉ, Phillips C. Cryo-EM: The Resolution Revolution and Drug Discovery. SLAS Discov. 2021 Jan;26(1):17-31. doi: 10.1177/2472555220960401. Epub 2020 Oct 5. PMID: 33016175.
