FDA Approval Roundup - December 2019
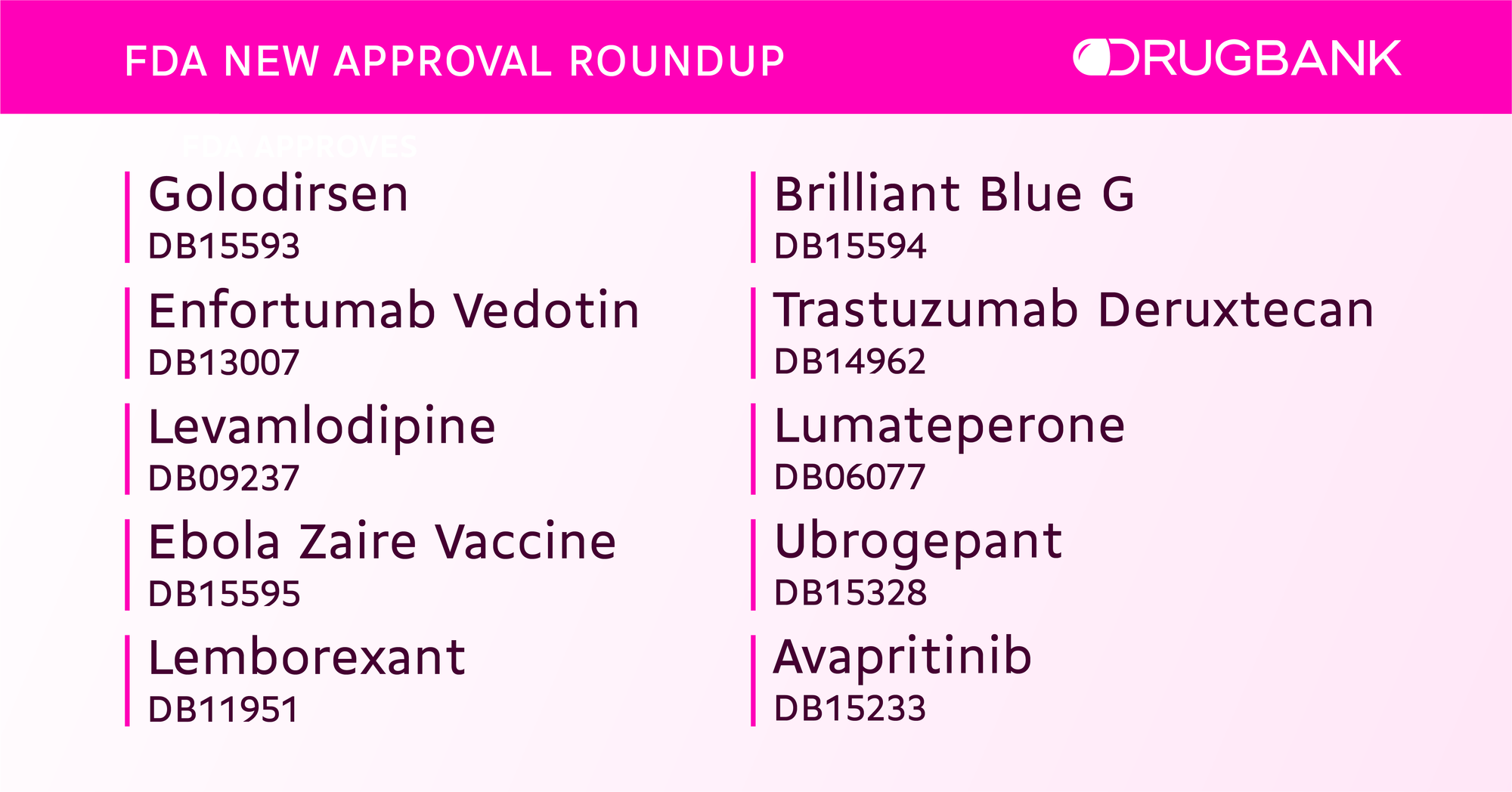
Last month the FDA approved ten new drugs aimed at treating a broad spectrum of diseases and ailments. The FDA often approves a number of drugs over a short period of time before the new year, with the group of approvals being aptly called "December Drugs". Check out the approvals below, and head to DrugBank.ca to learn more about them:
Dec 12, 2019 Golodirsen (DB15593)
FDA grants accelerated and conditional approval for Golodirsen, a drug for the treatment of Duchenne Muscular Dystrophy.
Drugbank.ca/drugs/DB15593
Dec 18, 2019 Enfortumab Vedotin (DB13007)
FDA grants accelerated approval to enfortumab vedotin, an antibody-drug conjugate for treatment-resistant urothelial cancers.
DrugBank.ca/drugs/DB13007
Dec 19, 2019 Levamlodipine (DB09237)
FDA approves levamlodipine, the pharmacologically active component of amlodipine.
DrugBank.ca/drugs/DB09237
Dec 19, 2019 Ebola Zaire Vaccine (DB15595)
FDA approves a live attenuated vaccine for the prevention of Zaire ebolavirus.
DrugBank.ca/drugs/DB15595
Dec 20, 2019 Lemborexant (DB11951)
FDA approves lemborexant, a dual orexin receptor antagonist for the treatment of insomnia.
DrugBank.ca/drugs/DB11951
Dec 20, 2019 Brilliant Blue G (DB15594)
FDA approves Brilliant Blue G for staining of the internal limiting membrane (ILM) of the eye.
Drugbank.ca/drugs/DB15594
Dec 20, 2019 Trastuzumab deruxtecan (DB14962)
FDA grants accelerated and conditional approval for Trastuzumab deruxtecan, a drug for the treatment of HER2 positive breast cancer.
Drugbank.ca/drugs/DB14962
Dec 20, 2019 Lumateperone (DB06077)
FDA approved Lumateperone, a second generation antipsychotic for the treatment of schizophrenia in adults.
DrugBank.ca/drugs/DB06077
Dec 23, 2019 Ubrogepant (DB15328)
FDA approves ubrogepant, the first oral CGRP antagonist approved for the acute treatment of migraines.
DrugBank.ca/drugs/DB15328
Jan 9, 2020 Avapritinib (DB15233)
FDA approves avapritinib, a selective tyrosine kinase inhibitor to treat unresectable, metastatic gastrointestinal stromal tumors.
DrugBank.ca/drugs/DB15233
